Volume 10, Issue 2 (2022)
Health Educ Health Promot 2022, 10(2): 247-253 |
Back to browse issues page
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Jawed S, Al-Taher Q, Shihab A. Melanoides tuberculate as Bioindicator of the Heavy Metal Contamination in Water and Sediment Pollution of Euphrates River at Thi-Qar province, Iraq. Health Educ Health Promot 2022; 10 (2) :247-253
URL: http://hehp.modares.ac.ir/article-5-61499-en.html
URL: http://hehp.modares.ac.ir/article-5-61499-en.html
1- Department of Biology, College of Education for Pure Sciences, University of Thi-Qar, Thi-Qar, Iraq
2- Department of biology, College of Basic Education, University of Sumer, Thi-Qar, Iraq
3- Ministry of Education, Thi-Qar, Iraq
2- Department of biology, College of Basic Education, University of Sumer, Thi-Qar, Iraq
3- Ministry of Education, Thi-Qar, Iraq
Full-Text [PDF 718 kb]
(4047 Downloads)
| Abstract (HTML) (2702 Views)
Full-Text: (797 Views)
Introduction
Water is one of the renewable sources that represent the base stone of the sustainability of human life and all kinds of living creatures on the surface of the earth [1]. The total water on the Earth's surface represents about 71%. Available freshwater represents 3%, of which available for human use constitutes only 0.4% [2]. Water is home to many trace minerals found in the oceans, where more than 70 elements have been confirmed [3]. The interest in studying the physical and chemical factors of aquatic environments, including rivers, is due to the possibility of these factors in these ecosystems create suitable conditions for the biological diversity of living organisms and the extent of changes that occurred due to human activity them from releases of organic carbon, nutrients and harmful substances, and their content of dissolved oxygen (DO) in them [4].
Heavy metals from anthropogenic pollution sources are always ejected into aquatic and terrestrial ecosystems therefore, the concern was increased to evaluate the effect of man pollution on a different kind of ecosystem is growing. Pollution with heavy metals is a hazard because of their poisonousness, bioaccumulation, and bio magnifications in the food chain [5]. Increasing heavy metals concentration can cause harmful and direct physiological toxic damage when they are deposited in tissues. Heavy metals are categorized into essential and non-essential groups and these groups convert to a toxic state at high intakes level [6]. The dispersal of industrialization, increase of population, and using serval technical devices of agriculture, have presented various pollutants into the aquatic ecosystems [7]. Heavy metals are released from manufacturing and transferred into the environment then concentrate and bio-amplified along the food chain [8].
The aquatic environment has a critical role in the dispersal and installation of living organisms communities signified by fish, macroinvertebrates, and other aquatic organisms [9].
The biodiversity of freshwater mollusks amounts to nearly 5,000 species, and Freshwater gastropods include about 4, 000 species [10], while the number of bivalves is approximately 1200 species [11].
Mollusks are a food source for fish, birds, and other invertebrates, and humans use them as a food source in many countries. Mollusks have a role in cycling the elements in nature and purifying water, so the presence of mollusks is a good vital indicator of environmental quality in all aquatic habitats [12]. Although mollusks are a food source for humans, they may pose a danger to them through the ability of these invertebrates to accumulate toxins inside their bodies in certain circumstances and thus expose humans to poisoning as a result of their consumption [13].
Melanoides tuberculata is famous as one of two universally invasive gastropods and it spreads through East Africa, the Middle East, and Southeast Asia. Melanoides tuberculata naturally lives in a wide diversity of freshwater or brackish water including rivers, streams, and wetlands [14, 15]. it is a cosmopolitan species from the class Gastropoda which has light brown color and elongated ¨ shells higher than wide, and it is a cosmopolitan species [16]. The snail has an operculum that can guard snail from dryness. This snail feeds mainly on microalgae and performances as an intermediate host for many trematodes [17].
It is a considerable food source for fish, some small carnivores, and water birds [18, 19], Moreover, they are intermediate predictors of many species of parasites that infect humans and animals. Furthermore, Melanoides tuberculata has features like a high growth rate, the capacity for migration, and territorial expansion. M. tuberculata is the greatest wide-ranging species of the family Thiaridae, which can live in practically any sort of freshwater causing this species one of the most persistent macroinvertebrates [20].
The current study aimed to determine and evaluated the heavy metals in water, sediment, and Melanoides tuberculata and their relationship with some physical and chemical factors.
Material and Methods
In this experimental research, the trace metal content (Cadmium: Cd, Lead: Pb, Nickle: Ni, Zink: Zn) was investigated in water, sediment, and the soft tissue and shell of Melanoides tuberculate. The Euphrates river is the lengthiest in Western Asia, the river initiates from Turkey and transfers to Syria, then arrives in Iraq and connects with the Tigris river in Basra city southern Iraq. The river is about 2786 km [21]. Four stations in the Euphrates river in Nasiriyah city were studied.
Water is one of the renewable sources that represent the base stone of the sustainability of human life and all kinds of living creatures on the surface of the earth [1]. The total water on the Earth's surface represents about 71%. Available freshwater represents 3%, of which available for human use constitutes only 0.4% [2]. Water is home to many trace minerals found in the oceans, where more than 70 elements have been confirmed [3]. The interest in studying the physical and chemical factors of aquatic environments, including rivers, is due to the possibility of these factors in these ecosystems create suitable conditions for the biological diversity of living organisms and the extent of changes that occurred due to human activity them from releases of organic carbon, nutrients and harmful substances, and their content of dissolved oxygen (DO) in them [4].
Heavy metals from anthropogenic pollution sources are always ejected into aquatic and terrestrial ecosystems therefore, the concern was increased to evaluate the effect of man pollution on a different kind of ecosystem is growing. Pollution with heavy metals is a hazard because of their poisonousness, bioaccumulation, and bio magnifications in the food chain [5]. Increasing heavy metals concentration can cause harmful and direct physiological toxic damage when they are deposited in tissues. Heavy metals are categorized into essential and non-essential groups and these groups convert to a toxic state at high intakes level [6]. The dispersal of industrialization, increase of population, and using serval technical devices of agriculture, have presented various pollutants into the aquatic ecosystems [7]. Heavy metals are released from manufacturing and transferred into the environment then concentrate and bio-amplified along the food chain [8].
The aquatic environment has a critical role in the dispersal and installation of living organisms communities signified by fish, macroinvertebrates, and other aquatic organisms [9].
The biodiversity of freshwater mollusks amounts to nearly 5,000 species, and Freshwater gastropods include about 4, 000 species [10], while the number of bivalves is approximately 1200 species [11].
Mollusks are a food source for fish, birds, and other invertebrates, and humans use them as a food source in many countries. Mollusks have a role in cycling the elements in nature and purifying water, so the presence of mollusks is a good vital indicator of environmental quality in all aquatic habitats [12]. Although mollusks are a food source for humans, they may pose a danger to them through the ability of these invertebrates to accumulate toxins inside their bodies in certain circumstances and thus expose humans to poisoning as a result of their consumption [13].
Melanoides tuberculata is famous as one of two universally invasive gastropods and it spreads through East Africa, the Middle East, and Southeast Asia. Melanoides tuberculata naturally lives in a wide diversity of freshwater or brackish water including rivers, streams, and wetlands [14, 15]. it is a cosmopolitan species from the class Gastropoda which has light brown color and elongated ¨ shells higher than wide, and it is a cosmopolitan species [16]. The snail has an operculum that can guard snail from dryness. This snail feeds mainly on microalgae and performances as an intermediate host for many trematodes [17].
It is a considerable food source for fish, some small carnivores, and water birds [18, 19], Moreover, they are intermediate predictors of many species of parasites that infect humans and animals. Furthermore, Melanoides tuberculata has features like a high growth rate, the capacity for migration, and territorial expansion. M. tuberculata is the greatest wide-ranging species of the family Thiaridae, which can live in practically any sort of freshwater causing this species one of the most persistent macroinvertebrates [20].
The current study aimed to determine and evaluated the heavy metals in water, sediment, and Melanoides tuberculata and their relationship with some physical and chemical factors.
Material and Methods
In this experimental research, the trace metal content (Cadmium: Cd, Lead: Pb, Nickle: Ni, Zink: Zn) was investigated in water, sediment, and the soft tissue and shell of Melanoides tuberculate. The Euphrates river is the lengthiest in Western Asia, the river initiates from Turkey and transfers to Syria, then arrives in Iraq and connects with the Tigris river in Basra city southern Iraq. The river is about 2786 km [21]. Four stations in the Euphrates river in Nasiriyah city were studied.
1.Station 1 is located at the beginning of the river when entering Nasiriyah city.
2.Station 2 is located near the thermal electric power station in the junction zone of hot water emerging with the river.
3.Station 3 is located far away from the second station when the river leaves the city of Al-Nasiriyah.
Measurements were made for all study sites for only two different seasons in terms of climatic conditions, the winter and summer seasons. The samples were collected using clean polyethylene bottles (Glaswarenfabrik Karl Hecht; Germani). Sampling was analyzed for some physical properties and checked immediately after collection. The air and water temperature were measured using a graduated mercury thermometer U14295 (3B Scientific GmbH; USA) from 0-100°C, while the pH concentration and electrical conductivity (EC; µS/cm1) were measured using a Multimeter device (EC-PCSTestr35; Hana Inc). In addition, DO was measured using the method described in a study [22]. The mollusk samples were also collected using a manual sieve with a diameter of 0.5mm, where the collected samples are washed with water to get rid of the impurities stuck in them, after which they are diagnosed and classified according to studies [23, 24]. Sediment samples, an exact weight of the dry sediment sample (0.5g) was fully digested in Teflon vessels using a mixture of concentrated acids (HNO3 and HCLO4 (AnalaR; Australia) for about 2 hours (triplicate digestion was made for each sample). The final solution was diluted to 25 ml with distilled de-ionized water. Their metal content was measured by AAS described by [25].
Sediments were analyzed by collecting samples using cleaned and acid-washed PVC corer and immediately kept in pre-cleaned and acid-washed polythene bags, which were sealed and kept in an ice box until further analysis in the laboratory. Sediment samples were washed with metal-free double distilled water. The sediment samples were then dried in an oven at 60°C for 5-6 hours. Dried sediment samples were ground in a glass mortar and reduced into fine particles. Metals were analyzed through atomic absorption spectrophotometer; 2.0g from soft tissues were weighed for mollusk then 1ml of digestion solution (concentrated nitric acid, concentrated sulfuric acid, and concentrated perchloric acid, with a percentage of 1:1:1) was added to flasks of content. The flasks closed strongly and left for 48-72 hours at room temperature to finish the digestion process. Then, completed to 10ml with deionized water and filtered. Finally, measured Atomic Spectrophotometric Absorption (Byahut Scientico; Iniamart; India).
Findings
Some physical and chemical parameters and heavy metals concentrations for water were studied in Tables 1 and 2. The result of the present study showed that the maximum mean concentration was reordered in summer and the minimum was in winter, the temperature of the air was higher than water, and station 2 recorded a high value for both air and water temperature. For pH, all the mean showed alkaline nature, and the average was arranged between 7.80-8. 40 and slight fluctuations in the values of pH appeared in along the study period. The EC mean in the aquatic location ranged from 1945.00-3319.00µS/cm-1. The highest mean was recorded in winter at station 2 whereas the lowest mean was recorded in summer at station 3. For DO mean concentration was arranged from 5.00- 7.12. Winter has recorded the highest mean at stations 2 and 3 while the lowest mean was recorded in summer at station 1.
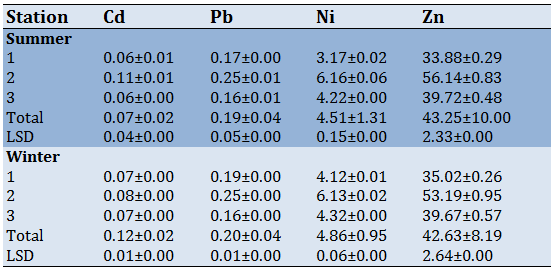
The mean concentration for some heavy metals in water was recorded (Table 2). The lowest and highest mean concentration was arranged between 0.06-0.11, 0.16-0.25, 3.17-6.16, 33.88-56.14µg/l for Cd, Pb, Ni, and Zn metals respectively. The result showed that station2 recorded the highest value of the mean concentration of metals. The descending order of heavy metals was: Zn.
Table 1) Mean results of some physical and chemical parameters for water in studied stations of Euphrates river at Thi-Qar
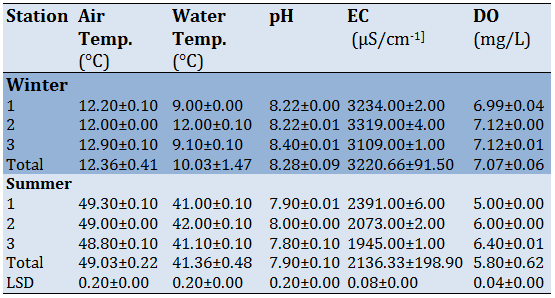
Table 2) Mean concentration of heavy metals (µg/l) for water in Euphrates river at Thi-Qar proven
The results of Table 3 showed the mean concentration and the stander deviation for heavy metals in sediments. The mean value in sediments ranged between 0.11-0.14, 8.16-10.49, 45.73-65.76, and 37.87-59.19µg/g for Cd, Pb, Ni, and Zn metals, respectively. The metals were arranged in order: Zn
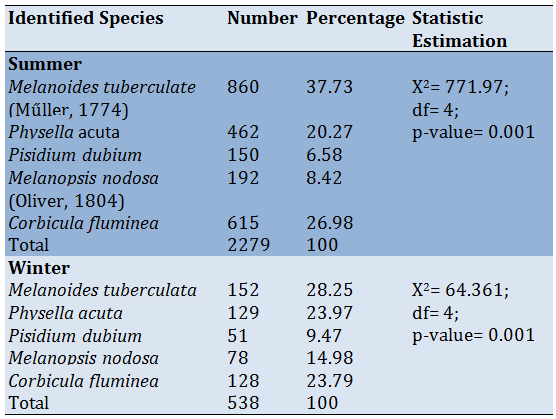
The highest frequency of the different species of mollusks in studied stations was recoded for Melanoides tuberculate, Corbicula fluminea, and Physella acuta, respectively; While the lowest frequency was for Melanopsis nodosa and Pisidium dubium in summer and winter, respectively. Melanoides tuberculate more dispersal and had the highest value in summer in the studied area (Table 4).
Table 3)Mean concentration of heavy metals (µg/g) for sediment in Euphrates river at Thi-Qar proven

Table 4) Statistic estimation of the aquatic Gastropod from three stations in Euphrates river at Thi-Qar proven
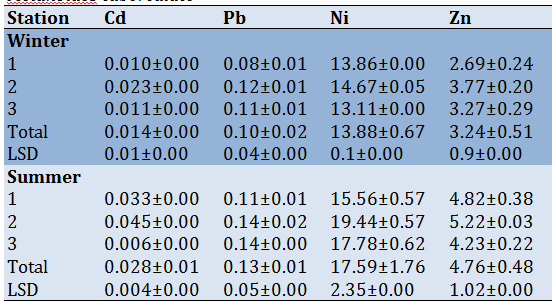
The mean concentration and slandered deviation for heavy metals in soft tissue and shell of Melanoides tuberculate showed in tables 5 and 6. Results revealed that the mean concentration of heavy metals was higher in soft tissue than in shells during the summer season. The mean concentration of Pb was high about 0.40-014µg/g in station 2 during summer in soft tissue and shell respectively; while Zn was 29.14-5.22 in station 2 during summer. The metals were arranged in order: Pb
Table 5) Mean concentration of heavy metals (µg/g) for soft tissue in Melanoides tuberculate
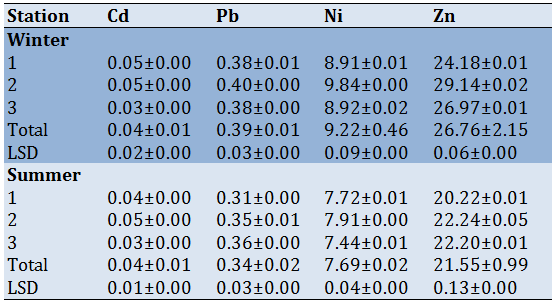
Table 6)Mean concentration of heavy metals (µg/g) for shell in Melanoides tuberculate
Discussion
The physical and chemical parameters of water are an essential factor for living the aquatic organism and affect their distribution of them [26]. Temperature is the most important factor for the living organism and influenced many processes like reproduction and distribution in both animals and plants [27]. The results showed that the temperature of the water was less than air, and water in both seasons cause they were affected by solar radiation from the sun and the water was shallow in that area [28]. That relationship made great conjunction between air and water temperature. For pH, the average was arranged between 7.80-8.40 which made the water slightly alkaline. Iraq water was tend to be alkaline [29]. The alkane was formed by the photosynthetic rate which depended on respiration and decomposition [30]. The electrical conductivity in the aquatic location is related to the soluble solids in the water therefore, it formed an excellent parameter to appear the purity of the water [31]. The value of electrical conductivity was 3220.66 in winter and 2136.33 in summer. The rising of the value in winter was related to the autumn rain during winter which increasing the washing of soil near the river.
The DO ratio was arranged from 5.00-7.12 and the DO value was high in winter than summer season because the increase in temperature decreases the DO. The high level of DO appears of good water quality [26]. Many factors affect the DO value like temperature and salinity and the number of abiotic [32].
Heavy metals are one of the more serious pollutants in our natural environment due to their toxicity, persistence, and bioaccumulation problems [33]. Heavy metals are pollutants that can yield huge harm to the atmosphere once they have high concentrations [19–34].
The present study showed clear differences in heavy metal concentrations between studied stations and seasons. The concentrations of heavy metals were higher in summer at station 3. This was due to the exposure of station 3 to several forms of contamination such as chemicals, waste, oil spilled, and sewage that were thrown from the thermal electric power station. Besides the presence of residential areas near this station.
Moreover, there were higher concentrations of studied metals in the summer than in the winter. This happened because of water evaporation which results from increasing temperatures and then increasing concentrations of heavy metals. Furthermore, degradation of organic matter could increase the heavy metals concentration [35] these results were agreed with studies [36-38].
Sediment formed an essential part of the river, it made a sink for it and most heavy metals settled on it. The current study revealed that sediment has a high concentration of heavy metals than water table 3. The results of the study were shown that the concentration of studied metals in sediment was higher than their concentration in water except for Pb. This happened due to depose of metals with the suspended matter in surface water at the bottom and increased with the ability of organic matter to adsorption [39, 40]. Station 2 has a high concentration of heavy metals compared to the other stations because of its location near the population area which throws the waste that increased the organic matter and then increases the absorption of heavy metals. Many factors affect the concentration of heavy metals in sediments such as pH, degradation of plankton, Diatoms, and organisms. More ever, temperature, salinity, the proportion of organic matter in sediments, and sediment grain size were the most important factor that affects the concentration of heavy metals in sediment [41]. The result agreed with [42, 43]. The present study showed a high concentration of Pb at station 2 in the summer is related to the high traffic density near this area of Al-Nassiriya city.
The result in table 4 was showed the percentage of the different species of mollusks in the studied stations. The highest presence was recorded for Melanoides tuberculate (37.7-152%), Corbicula fluminea (26.98-128%) then Physella acuta (20.27-23.78%) while the lowest parentages were Melanopsis nodosa (8.2-1.98%) and Pisidium dubium (6.58-9.47%) for summer and winter respectively. The current study showed an increase in the appearance of some species during summer more than winter cause the increasing temperature could increase the metabolic activity of Mollusca. Furthermore, the physical and chemical parameters for water affect the growth and diversity of aquatic invertebrates [44]. Melanoides tuberculate has the highest ratio in the Euphrates river cause these species prefer the lotic water. All studied species were found during both seasons and this disagrees with research that showed that many species of Mollusca die in the winter or low-temperature degrees [45, 46]. Interestingly, the rising temperature formed an essentially important part in the proliferation of snails. Our study is constant with the study [29].
The present study revealed that the concentration of heavy metals in snail tissue and the shell was higher in summer than in winter at station 2 Table [4, 5]. Interestingly the concentration of Pb and Ni in tissue was less than in the shell. It is well known that the uptake of trace metals by an aquatic organism is dependent on the metal concentration in the solution until situated [47]. Therefore, the heavy metals concentration in the snail was more than in water cause the organic matter keep the metals the snail absorb. The level of accumulation in the organism was changed according to the ambient concentration and environmental concentrations of metals [48].
The Pb concentration in the studied snail was high epically in sediment and soft tissue which exceeded the presumable level and made 0.30 mg/kg set by [49]. The high concentration can produce a possible risk of toxicity and Pb poisoning. Zn is a vital nutrient for all organisms. The concentrations of Zn were high in all studied samples but within the FAO [50] reference limit of 130.0 mg/kg. increasing of intake Zn from organisms having a high concentration of Zn resulting in vomiting, nausea, headaches, and diarrhea. Inflowing of wastewater from the thermal station and area near station 2 was the main source of raising the concentration of Zn. The result of the present study agreed with [51, 52]. Al most of the heavy metal concentrations exceed the permissible level of WHO [53].
Conclusion
This study declared that the concentration of heavy metals concentration is elevated in the specific station which has waste from human activities and these added levels of metals to the river. So attention should increase to monitoring the level of heavy metals and use organisms like snails and shrimp as bio-indicator.
Acknowledgments: None declared.
Ethical Permissions: None declared.
Conflicts of Interests: None declared.
Authors’ Contributions: Jawed S.T (First Author), Introduction Writer (30%); Al-Taher Q.M (Second Author), Methodologist/Main Researcher/Discussion Writer (40%); Shihab A.F (Third Author), Assistant Researcher/Discussion Writer (30%)
Funding/Support: None declared.
Sediments were analyzed by collecting samples using cleaned and acid-washed PVC corer and immediately kept in pre-cleaned and acid-washed polythene bags, which were sealed and kept in an ice box until further analysis in the laboratory. Sediment samples were washed with metal-free double distilled water. The sediment samples were then dried in an oven at 60°C for 5-6 hours. Dried sediment samples were ground in a glass mortar and reduced into fine particles. Metals were analyzed through atomic absorption spectrophotometer; 2.0g from soft tissues were weighed for mollusk then 1ml of digestion solution (concentrated nitric acid, concentrated sulfuric acid, and concentrated perchloric acid, with a percentage of 1:1:1) was added to flasks of content. The flasks closed strongly and left for 48-72 hours at room temperature to finish the digestion process. Then, completed to 10ml with deionized water and filtered. Finally, measured Atomic Spectrophotometric Absorption (Byahut Scientico; Iniamart; India).
Findings
Some physical and chemical parameters and heavy metals concentrations for water were studied in Tables 1 and 2. The result of the present study showed that the maximum mean concentration was reordered in summer and the minimum was in winter, the temperature of the air was higher than water, and station 2 recorded a high value for both air and water temperature. For pH, all the mean showed alkaline nature, and the average was arranged between 7.80-8. 40 and slight fluctuations in the values of pH appeared in along the study period. The EC mean in the aquatic location ranged from 1945.00-3319.00µS/cm-1. The highest mean was recorded in winter at station 2 whereas the lowest mean was recorded in summer at station 3. For DO mean concentration was arranged from 5.00- 7.12. Winter has recorded the highest mean at stations 2 and 3 while the lowest mean was recorded in summer at station 1.
The mean concentration for some heavy metals in water was recorded (Table 2). The lowest and highest mean concentration was arranged between 0.06-0.11, 0.16-0.25, 3.17-6.16, 33.88-56.14µg/l for Cd, Pb, Ni, and Zn metals respectively. The result showed that station2 recorded the highest value of the mean concentration of metals. The descending order of heavy metals was: Zn
Table 1) Mean results of some physical and chemical parameters for water in studied stations of Euphrates river at Thi-Qar

Table 2) Mean concentration of heavy metals (µg/l) for water in Euphrates river at Thi-Qar proven

The results of Table 3 showed the mean concentration and the stander deviation for heavy metals in sediments. The mean value in sediments ranged between 0.11-0.14, 8.16-10.49, 45.73-65.76, and 37.87-59.19µg/g for Cd, Pb, Ni, and Zn metals, respectively. The metals were arranged in order: Zn
The highest frequency of the different species of mollusks in studied stations was recoded for Melanoides tuberculate, Corbicula fluminea, and Physella acuta, respectively; While the lowest frequency was for Melanopsis nodosa and Pisidium dubium in summer and winter, respectively. Melanoides tuberculate more dispersal and had the highest value in summer in the studied area (Table 4).
Table 3)Mean concentration of heavy metals (µg/g) for sediment in Euphrates river at Thi-Qar proven

Table 4) Statistic estimation of the aquatic Gastropod from three stations in Euphrates river at Thi-Qar proven

The mean concentration and slandered deviation for heavy metals in soft tissue and shell of Melanoides tuberculate showed in tables 5 and 6. Results revealed that the mean concentration of heavy metals was higher in soft tissue than in shells during the summer season. The mean concentration of Pb was high about 0.40-014µg/g in station 2 during summer in soft tissue and shell respectively; while Zn was 29.14-5.22 in station 2 during summer. The metals were arranged in order: Pb
Table 5) Mean concentration of heavy metals (µg/g) for soft tissue in Melanoides tuberculate

Table 6)Mean concentration of heavy metals (µg/g) for shell in Melanoides tuberculate

Discussion
The physical and chemical parameters of water are an essential factor for living the aquatic organism and affect their distribution of them [26]. Temperature is the most important factor for the living organism and influenced many processes like reproduction and distribution in both animals and plants [27]. The results showed that the temperature of the water was less than air, and water in both seasons cause they were affected by solar radiation from the sun and the water was shallow in that area [28]. That relationship made great conjunction between air and water temperature. For pH, the average was arranged between 7.80-8.40 which made the water slightly alkaline. Iraq water was tend to be alkaline [29]. The alkane was formed by the photosynthetic rate which depended on respiration and decomposition [30]. The electrical conductivity in the aquatic location is related to the soluble solids in the water therefore, it formed an excellent parameter to appear the purity of the water [31]. The value of electrical conductivity was 3220.66 in winter and 2136.33 in summer. The rising of the value in winter was related to the autumn rain during winter which increasing the washing of soil near the river.
The DO ratio was arranged from 5.00-7.12 and the DO value was high in winter than summer season because the increase in temperature decreases the DO. The high level of DO appears of good water quality [26]. Many factors affect the DO value like temperature and salinity and the number of abiotic [32].
Heavy metals are one of the more serious pollutants in our natural environment due to their toxicity, persistence, and bioaccumulation problems [33]. Heavy metals are pollutants that can yield huge harm to the atmosphere once they have high concentrations [19–34].
The present study showed clear differences in heavy metal concentrations between studied stations and seasons. The concentrations of heavy metals were higher in summer at station 3. This was due to the exposure of station 3 to several forms of contamination such as chemicals, waste, oil spilled, and sewage that were thrown from the thermal electric power station. Besides the presence of residential areas near this station.
Moreover, there were higher concentrations of studied metals in the summer than in the winter. This happened because of water evaporation which results from increasing temperatures and then increasing concentrations of heavy metals. Furthermore, degradation of organic matter could increase the heavy metals concentration [35] these results were agreed with studies [36-38].
Sediment formed an essential part of the river, it made a sink for it and most heavy metals settled on it. The current study revealed that sediment has a high concentration of heavy metals than water table 3. The results of the study were shown that the concentration of studied metals in sediment was higher than their concentration in water except for Pb. This happened due to depose of metals with the suspended matter in surface water at the bottom and increased with the ability of organic matter to adsorption [39, 40]. Station 2 has a high concentration of heavy metals compared to the other stations because of its location near the population area which throws the waste that increased the organic matter and then increases the absorption of heavy metals. Many factors affect the concentration of heavy metals in sediments such as pH, degradation of plankton, Diatoms, and organisms. More ever, temperature, salinity, the proportion of organic matter in sediments, and sediment grain size were the most important factor that affects the concentration of heavy metals in sediment [41]. The result agreed with [42, 43]. The present study showed a high concentration of Pb at station 2 in the summer is related to the high traffic density near this area of Al-Nassiriya city.
The result in table 4 was showed the percentage of the different species of mollusks in the studied stations. The highest presence was recorded for Melanoides tuberculate (37.7-152%), Corbicula fluminea (26.98-128%) then Physella acuta (20.27-23.78%) while the lowest parentages were Melanopsis nodosa (8.2-1.98%) and Pisidium dubium (6.58-9.47%) for summer and winter respectively. The current study showed an increase in the appearance of some species during summer more than winter cause the increasing temperature could increase the metabolic activity of Mollusca. Furthermore, the physical and chemical parameters for water affect the growth and diversity of aquatic invertebrates [44]. Melanoides tuberculate has the highest ratio in the Euphrates river cause these species prefer the lotic water. All studied species were found during both seasons and this disagrees with research that showed that many species of Mollusca die in the winter or low-temperature degrees [45, 46]. Interestingly, the rising temperature formed an essentially important part in the proliferation of snails. Our study is constant with the study [29].
The present study revealed that the concentration of heavy metals in snail tissue and the shell was higher in summer than in winter at station 2 Table [4, 5]. Interestingly the concentration of Pb and Ni in tissue was less than in the shell. It is well known that the uptake of trace metals by an aquatic organism is dependent on the metal concentration in the solution until situated [47]. Therefore, the heavy metals concentration in the snail was more than in water cause the organic matter keep the metals the snail absorb. The level of accumulation in the organism was changed according to the ambient concentration and environmental concentrations of metals [48].
The Pb concentration in the studied snail was high epically in sediment and soft tissue which exceeded the presumable level and made 0.30 mg/kg set by [49]. The high concentration can produce a possible risk of toxicity and Pb poisoning. Zn is a vital nutrient for all organisms. The concentrations of Zn were high in all studied samples but within the FAO [50] reference limit of 130.0 mg/kg. increasing of intake Zn from organisms having a high concentration of Zn resulting in vomiting, nausea, headaches, and diarrhea. Inflowing of wastewater from the thermal station and area near station 2 was the main source of raising the concentration of Zn. The result of the present study agreed with [51, 52]. Al most of the heavy metal concentrations exceed the permissible level of WHO [53].
Conclusion
This study declared that the concentration of heavy metals concentration is elevated in the specific station which has waste from human activities and these added levels of metals to the river. So attention should increase to monitoring the level of heavy metals and use organisms like snails and shrimp as bio-indicator.
Acknowledgments: None declared.
Ethical Permissions: None declared.
Conflicts of Interests: None declared.
Authors’ Contributions: Jawed S.T (First Author), Introduction Writer (30%); Al-Taher Q.M (Second Author), Methodologist/Main Researcher/Discussion Writer (40%); Shihab A.F (Third Author), Assistant Researcher/Discussion Writer (30%)
Funding/Support: None declared.
Article Type: Original Research |
Subject:
Health Education and Health Behavior
Received: 2022/03/30 | Accepted: 2022/05/25 | Published: 2022/06/12
Received: 2022/03/30 | Accepted: 2022/05/25 | Published: 2022/06/12
References
1. Wei SK, Gnauck A. Simulating water conflicts using game theoretical models for water resources management. Ecosyst Sustain Dev VI. 2007;106:3-12. [Link] [DOI:10.2495/ECO070011]
2. Gogoi BJ. Water scarcity: a major concern for citizens. Int J Civil Eng Technol. 2019;10(6):43-53. [Link]
3. Hou CW, Tsai YS, Jean WH, Chen CY, Ivy JL, Huang CY, et al. Deep ocean mineral water accelerates recovery from physical fatigue. J Int Soc Sports Nutr. 2013;10(1):7. [Link] [DOI:10.1186/1550-2783-10-7]
4. Meybeck M. Chemical characteristics of rivers. Fresh Surf Water. 2009;1:310. [Link]
5. Demirezen O, Uruc K. Comparative study of trace elements in certain fish, meat and meat products. Meat Sci. 2006;74(2):255-60. [Link] [DOI:10.1016/j.meatsci.2006.03.012]
6. Khurshid SJ, Qureshi IH. The role of inorganic elements in human body. Nucleus. 1984;21:3-23. [Link]
7. Ogbeibu AE, Ezeunara PU. Ecological impact of brewery effluent on Ikpoba river using the fish communities as bio- indicators. J Agua Res. 2002;17:35-44. [Link] [DOI:10.4314/jas.v17i1.19908]
8. Oguzie AE. Heavy metals in water and sediment of the lower Ikpoba river, Benin City, Nigeria. Sri Lanka J Aqua Sci. 2003;46(3):156-60. [Link]
9. Johnson PE. Sustaining Americas aquatic biodiversity-freshwater snail biodiversity and conservation. Virginia: VCE Publications; 2009. [Link]
10. Strong EE, Gargominy O, Ponder WF, Bouchet P. Global diversity of gastropods (Gastropoda; Mollusca) in freshwater. In: Balian EV, Lévêque C, Segers H, Martens K. Freshwater animal diversity assessment. Dordrecht: Springer; 2007. pp. 149-166. [Link] [DOI:10.1007/978-1-4020-8259-7_17]
11. Bogan AE. Global diversity of freshwater mussels (Mollusca, Bivalvia). In: Balian EV, Lévêque C, Segers H, Martens K. Freshwater animal diversity assessment. Dordrecht: Springer; 2007. pp. 139-147. [Link] [DOI:10.1007/978-1-4020-8259-7_16]
12. LAWAL-ARE AO, MORUF OR, JUNAID DA. Chemical bio-compounds and functional properties of raw and processed cuttlefish, Sepia officinalis (Mollusca: Cephalopoda). Food Environ Safe J. 2018;17(3). [Link]
13. Àvila C, Iken K, Fontana A, Cimino G. Chemical ecology of the Antarctic nudibranch Bathydoris hodgsoni Eliot, 1907: defensive role and origin of its natural products. J Exp Ma Biol Ecol. 2000;252(1):27-44. [Link] [DOI:10.1016/S0022-0981(00)00227-6]
14. Facon B, Pointier JP, Glaubrecht M, Poux C, Jarne P, David P. A molecular phylogeography approach to biological invasions of the New World by parthenogenetic thiarid snails. Mol Ecol. 2003;12(11): 3027-39. [Link] [DOI:10.1046/j.1365-294X.2003.01972.x]
15. Perissinotto R, Miranda NAF, Raw JL, Peer N. Biodiversity census of Lake St Lucia, iSimangaliso Wetland Park (South Africa): gastropod molluscs. ZooKeys. 2014;440:1-43. [Link] [DOI:10.3897/zookeys.440.7803]
16. Yong HS. Freshwater invertebrates of the Malaysian region. Kuala Lumpur: Academy of Science Malaysia; 2004. pp. 225-253. [Link]
17. Mitchell AJ, Hobbs MS, Brandt TM. The effect of chemical treatments on red-rim melania Melanoides tuberculata, an exotic aquatic snail that serves as a vector of trematodes to fish and other species in the USA. North Am J Fish Manag. 2007;27(4):1287-93. [Link] [DOI:10.1577/M06-252.1]
18. Hann BJ, Mundy CJ, Goldsborough LG. Snail-periphyton interactions in a prairie lacustrine wetland. Hydrobiologia. 2001;457(1):167-75. [Link] [DOI:10.1023/A:1012231601521]
19. Sitnikova T, Soldatenko E, Kamaltynov R, Riedel F. The finding of North American freshwater gastropods of the genus Planorbella Haldeman, 1842 (Pulmonata: Planorbidae) in East Siberia. Aqua Invasions. 2010;5(2):201-5. [Link] [DOI:10.3391/ai.2010.5.2.12]
20. Flores MJL, Zaffaralla M. Macroinvertebrate composition, diversity and richness in relation to the water quality status of Mananga River, Cebu, Philippines. Philippine Sci Lett. 2012;5(2):103-13. [Link]
21. Al-Ansari N, Adamo N, Sissakian V, Knutsson S, Laue J. Water resources of the Euphrates River catchment. J Earth Sci Geotechn Eng. 2018;8:1-20. [Link]
22. Gilcreas FW. Standard methods for the examination of water and waste water. Am J Public Health Nat Health. 1966;56(3):387-388. [Link] [DOI:10.2105/AJPH.56.3.387]
23. Al-Kandari M, Oliver PG, Chen W, Skryabin V, Raghu M, Yousif A, et al. Diversity and distribution of the intertidal Mollusca of the State of Kuwait, Arabian Gulf. Region Stud Mar Sci. 2020;33:100905. [Link] [DOI:10.1016/j.rsma.2019.100905]
24. Pennak RW. Fresh-Water Biology. Henry Baldwin Ward and George Chandler Whipple. WT Edmondson, Ed. Wiley, New York; Chapman and Hall, London, ed. 2, 1959. xx+ 1248 pp. Illus. $34.50. Science. 1960;131(3396):296. [Link] [DOI:10.1126/science.131.3396.296]
25. Usero J, Gonza'lez-Regalado E, Gracia I. Trace metals in the bivalve mollusc Chamelea gallina from the Atlantic coast of southern Spain. Mar Pollut Bull. 1996;32(3):305-10. [Link] [DOI:10.1016/0025-326X(95)00209-6]
26. Hays KB, White LD. Healthy range watersheds critical to the future of Texas. Texas Agricultural Extension Service. The Texas A&M University System. Water Texans Ser. 1998;(2). [Link]
27. Zlotorzynski A. The application of microwave radiation to analytical and environmental chemistry. Crit Rev Anal Chem. 1995;25(1):43-76. [Link] [DOI:10.1080/10408349508050557]
28. Corbitt RA. Standard handbook of environmental engineering. New York: McGraw-Hill; 1998. [Link]
29. ALKhafaji BY. Trace metals distribution in fish tissues (Cyprinus carpio and Barbus luteus) and sediments from Al-Msab Alamm River near the center of Al-Nassiriya city. Univ Thi-Qar J Sci. 2012;3(2):22-30. [Link]
30. Wani LA, Subla BA. Physic-Chemical features of two shallow Himalyan lakes. Bull Environ Sci. 1990;8:33-49. [Link]
31. Lembi CA. Limnology, lake and river ecosystems. J Physiol. 2001;37(6):1146-7. [Link] [DOI:10.1046/j.1529-8817.2001.37602.x]
32. ElNemaki F, Ali A, Zeinthom M, Radwan M. Impact of different water resources on the ecological parameters and the quality of Tilipia production at El-Abbassa fish farms in Egypt. 8th Int symposium on Tiliapa in Aquaculture. Unknown publisher; 2008. [Chines] [Link]
33. Tam NFY, Wong Y. Spatial variation of heavy metals in surface sediments of Hong Kong mangrove swamps. Environ Pollut. 2000;110(2):195-205. [Link] [DOI:10.1016/S0269-7491(99)00310-3]
34. Pyron M, Brown KM. Chapter 18 - Introduction to Mollusca and the Class Gastropoda. Ecol Gen Biol. 2015;383-421. [Link] [DOI:10.1016/B978-0-12-385026-3.00018-8]
35. Park N, Kim JH, Cho J. Organic matter, anion and metal wastewater treatment in Damyang surface flow constructed wetland in Kora. Ecol Eng. 2008;32(1):68-71. [Link] [DOI:10.1016/j.ecoleng.2007.09.003]
36. Al-Haidary MJS. Distribution of heavy metals in Mesopotamia marsh ecosystem, Iraq [dissertation]. Baghdad; University of Baghdad; 2009. [Arabic] [Link]
37. Muhsin AH. Pollution by heavy metals and bioaccumulation in Aquatic invertebrates in water surface in Basrah [dissertation]. Basrah: University of Basrah; 2011. [Arabic] [Link]
38. Radeef AF, Mosa AA, Qadir RM. Extraction and Estimation of some trace elements from different fish species from Tigris River, Kurdistan Region of Iraq. Tikrit J Pure Sci. 2013;18(3). [Link]
39. Elder JF. Metal biogeochemistry in surface water systems, a review of principles and concepts. Circular. 1989;1013. [Link] [DOI:10.3133/cir1013]
40. Yap CK, Ismail A, Tan SG & Omar H Accumulation, depuration and distribution of cadmium and zinc in the green-lipped mussel Perna viridis (Linnaeus) under laboratory conditions. Hydrobiologia 2000;498:151-60. [Link]
41. Bentivegna CS, Alfano JE, Bugel SM, Czechowicz K. Influence of sediment characteristics on heavy metal toxicity in an urban marsh. Urban Habitats. 2004;2(1):91-111. [Link]
42. Madkour HA. Distribution and relationship of heavy metal in the giant clam (Tridacana maxima) and associated sediments from different sites in the Egyptian Red sea coast. Egypt J Aqua Res. 2005;31(2):45-59. [Link]
43. Kazaar AA. Determination of some trace metals in environment and three types of Mollusca in Al-Hammar west [dissertation]. Basrah: University of Basrah; 2009. [Arabic] [Link]
44. Budha PB, Dutta J, Daniel BA. Bellamya bengalensis [Internet]. IUCN Red List of Threatened Species; 2013 [Unknown cited]. Available from: https://www.iucnredlist.org/species/166706/6265508 [Link]
45. Karimi GR, Derakhshanfar M, Pay Rari H. Population density. Trematodal Infection of Ecology of Lymnaea snail in Shadegan, Iran. Arch Razi Ins. 2004;58(1):125-9. [Link]
46. Neam NH, Al-Taee MM. Biodiversity of macroinvertebrates in Al-razzaza lake at Karbala/Iraq. Int J Adv Res. 2015;3(1):423-7. [Link]
47. Likens GE. Encyclopedia of inland waters. New York: Elsevier; 2009. [Link] [DOI:10.1016/B978-012370626-3.00001-6]
48. Mielke HW. Lead in the inner cities: Policies to reduce children's exposure to lead may be overlooking a major source of lead in the environment. Am Sci. 1999;87(1):62-73. [Link] [DOI:10.1511/1999.1.62]
49. European :union:. Commission Regulation (EC) No 629/2008 of 2 July 2008 amending Regulation (EC) No 1881/2006 setting maximum levels for certain contaminants in foodstuffs. Off J Eur :union:. 2008;173. [Link]
50. FAO. Compilation of legal limits for hazardous substances in fish and fishery products. Fisheries Circular. No. 764. Geneva: FAO; 1983. [Link]
51. Kesavan, K, Murugan, A, Venkatesan, V, Kumar, V. Heavy metal accumulation in molluscs and sediment from Uppanar estuary, southeast coast of India. Thalassas. 2013;29(2):15-21. [Link]
52. Salman JM, Randall AH, Ayad MJ, Almamoori. Seasonal variation of heavy metal in water and two species of Molluscs in Hilla River Iraq. Int J Geol Earth Environ Sci. 2002;4(2):16-24. [Link]
53. World Health Organization. Guidelines for drinking water. Geneva: World Health Organization; 2005. [Link]
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |





